MCAT General Chemistry Review Summary
Gold Standard MCAT General Chemistry Review Summary ('cheat sheet' notes)
MCAT Stoichiometry | Electronic Structure | Bonding | Phases | Solutions | Acids & Bases | Thermodynamics | Thermochemistry | Kinetics | Electrochemistry
Get the definitive MCAT General Chemistry Review Summary and conquer this section! This guide highlights the most high-yield topics, definitions, and essential equations required for a top score. Use this General Chemistry cheat sheet to strategically minimize rote memorization and maximize your study time. Need more practice? Sign up for our free MCAT practice test today, or see the full topic list.
Download the Gold Standard MCAT General Chemistry Summary PDF for FREE (no registration required).
MCAT General Chemistry Review: Stoichiometry
We'll break down the core concepts of mole ratios, chemical reactions, and reaction yields, equipping you with the skills to conquer stoichiometry problems with ease.
Note: Unless mentioned otherwise, the following images are excerpts from the Gold Standard MCAT General Chemistry ebook.
Mole - Atomic and Molecular Weights

Categories of Chemical Reactions
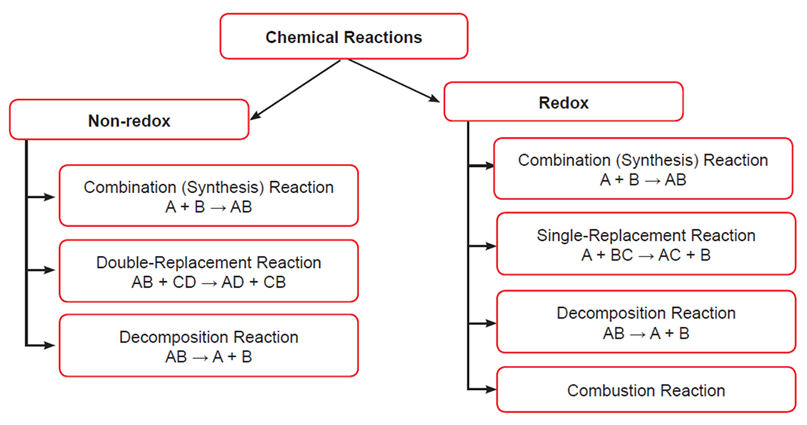
Note: Any reaction that does not involve the transfer of electrons (= change in oxidation numbers) qualifies as a non-redox reaction. Combination reactions qualify as non-redox reactions when all reactants and products are compounds and the oxidation numbers do not change. Decomposition reactions qualify as non-redox reactions when all reactants and products are compounds and the oxidation numbers do not change.
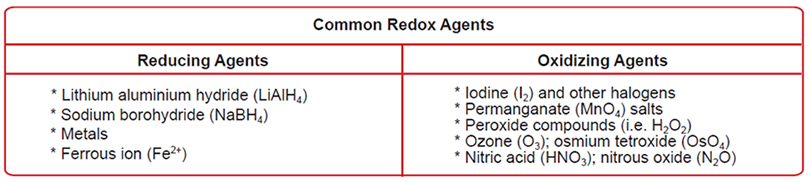
Oxidation Numbers, Redox Reactions, Oxidizing vs. Reducing Agents
- Here are the general rules:
- In elementary substances, the oxidation number of an uncombined element is zero
- In monatomic ions the oxidation number of the elements that make up this ion is equal to the charge of the ion
- In a neutral molecule the sum of the oxidation numbers of all the elements that make up the molecule is zero
- Some useful oxidation numbers to memorize
- For H: +1, except in metal hydrides where it is equal to -1
- For O: -2 in most compounds; In peroxides (e.g. in H2O2) the oxidation number for O is -1, it is +2 in OF2 and -1/2 in superoxides
- For alkali metals: +1
- For alkaline earth metals: +2
- Aluminium always has an oxidation number of +3 in all its compounds
- Here are the general rules:
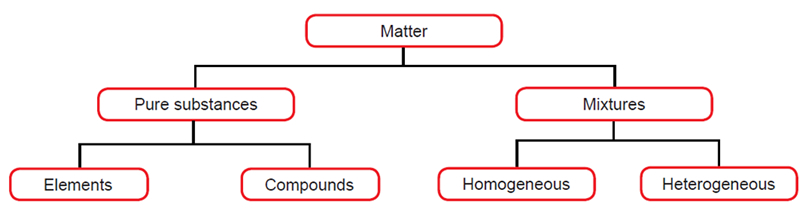
Mixtures
Don't practice on paper if the real exam doesn't use it. Master your scratch-work layout on our MCAT Erasable Noteboard and Marker —the exact dimensions and feel of the real thing.
Electronic Structure & The Periodic Table
- Conventional Notation for Electronic Structure
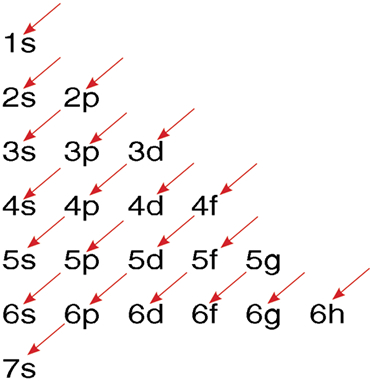
The order for filling atomic orbitals: Follow the direction of successive arrows moving from top to bottom.
Metals, Nonmetals and Metalloids
*General Characteristics of metals, nonmetals and metalloids
MCAT General Chemistry Review: Bonding
- Partial Ionic Character
-
This polar bond will also have a dipole moment given by:
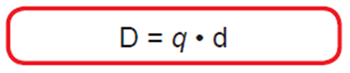
where q is the charge and d is the distance between these two atoms.
-
- Lewis Acids and Lewis Bases
-
The Lewis acid BF3 and the Lewis base NH3. Notice that the green arrows follow the flow of electron pairs.
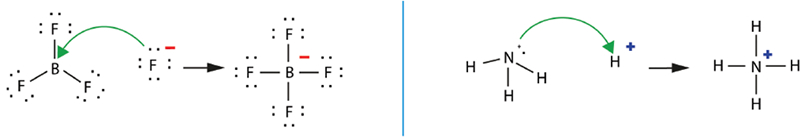
-
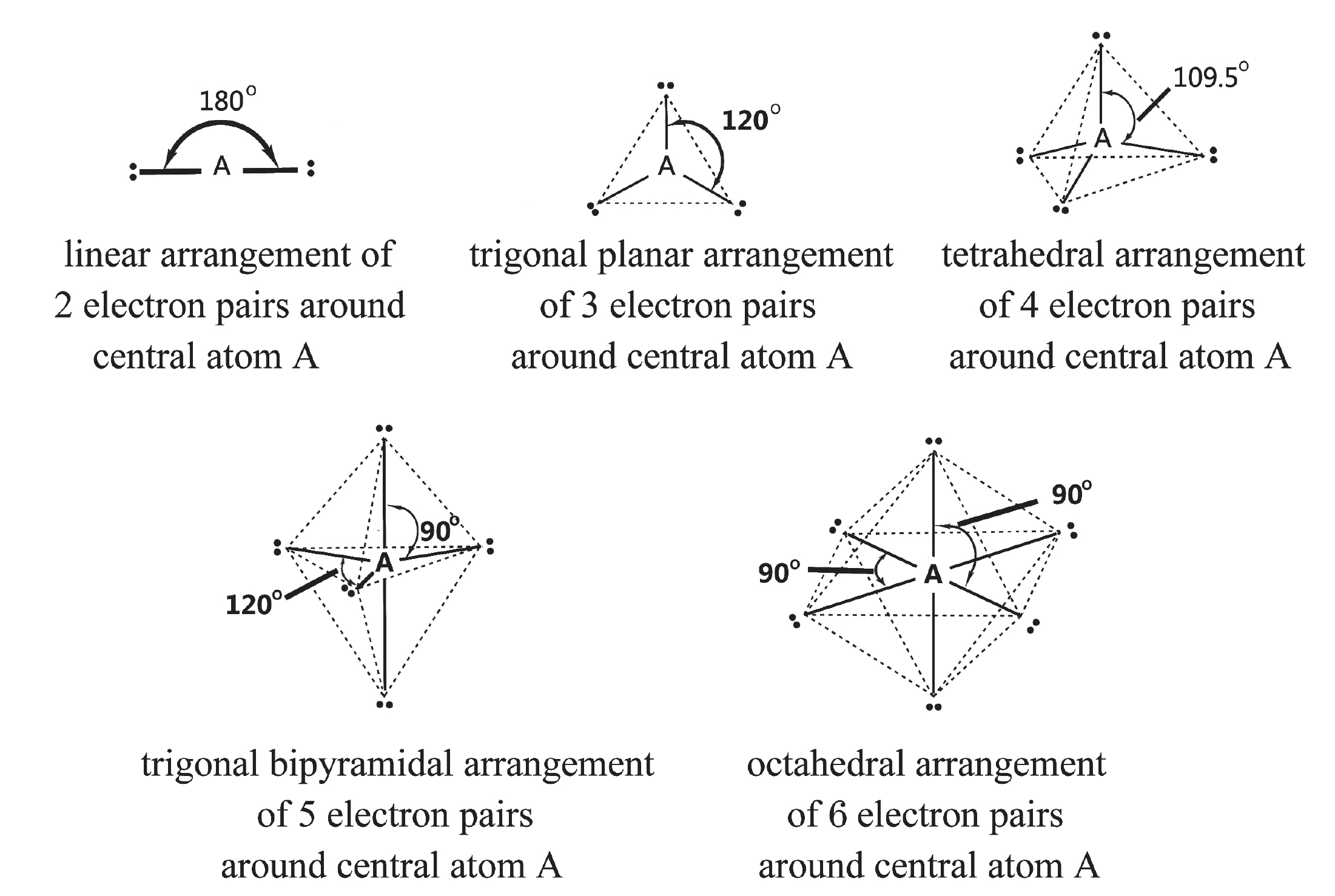
- Valence Shell Electronic Pair Repulsions (VSEPR Models)
-
Geometry of simple molecules in which the central atom A has one or more lone pairs of electrons (= e-)
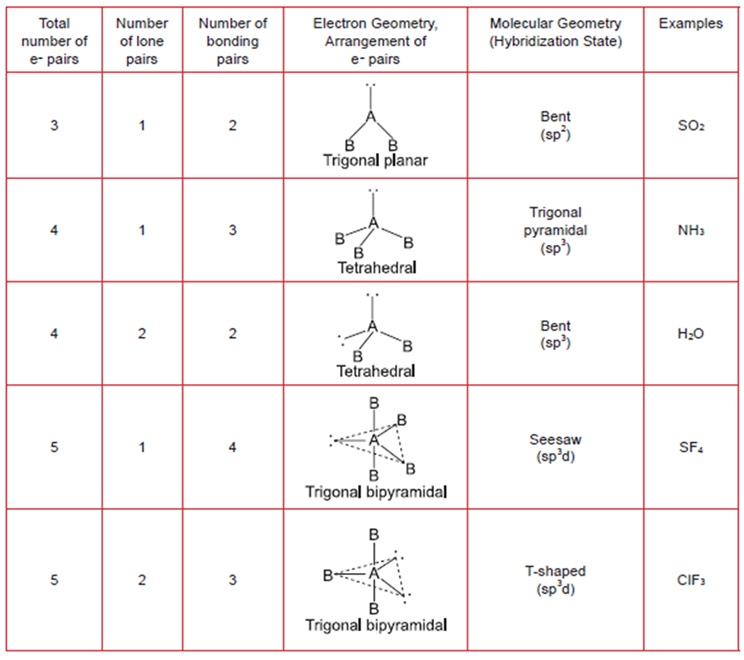
Note: dotted lines only represent the overall molecular shape and not molecular bonds. In brackets under "Molecular Geometry" is the hybridization as discussed in ORG 1.2.
-
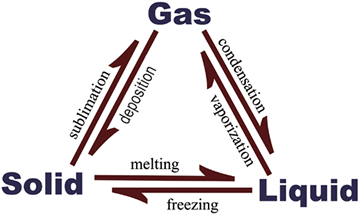
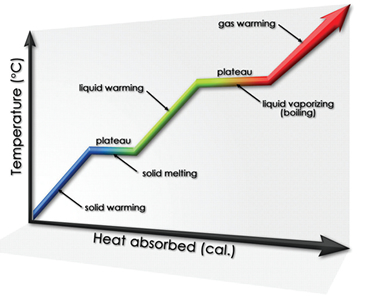
Phases and Phase Equilibria
This MCAT General Chemistry Review guide will take you on a journey through the different states of matter – solid, liquid, and gas.
- Standard Temperature and Pressure, Standard Molar Volume
- 0 °C (273.15 K) and 1.00 atm (101.33 kPa = 760 mmHg = 760 torr); these conditions are known as the standard temperature and pressure (STP). {Note: the SI unit of pressure is the pascal (Pa).}
- The volume occupied by one mole of any gas at STP is referred to as the standard molar volume and is equal to 22.4 L.
- Kinetic Molecular Theory of Gases (A Model for Gases)
- The average kinetic energy of the particles (KE = 1/2 mv2) increases in direct proportion to the temperature of the gas (KE = 3/2 kT) when the temperature is measured on an absolute scale (i.e. the Kelvin scale) and k is a constant (the Boltzmann constant).
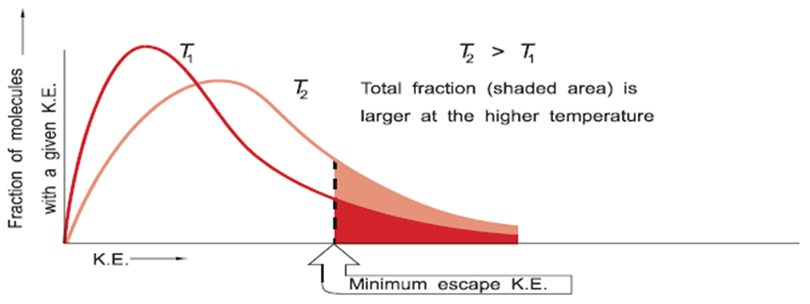
- The Maxwell Distribution Plot
Graham's Law (Diffusion and Effusion of Gases) 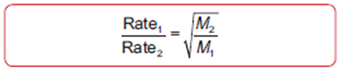
Combined Gas Law 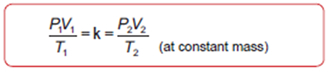
Charles' Law 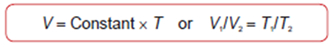
Ideal Gas Law 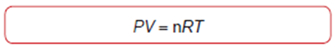
since m/V is the density (d) of the gas: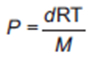
Boyle's Law 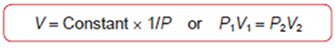
Partial Pressure and Dalton's Law Of course, the sum of all mole fractions in a mixture must equal one: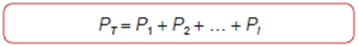
ΣX1 = 1Avogadro's Law 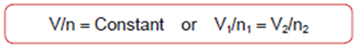
The partial pressure (Pi) of a component of a gas mixture is equal to: 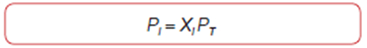
- The average kinetic energy of the particles (KE = 1/2 mv2) increases in direct proportion to the temperature of the gas (KE = 3/2 kT) when the temperature is measured on an absolute scale (i.e. the Kelvin scale) and k is a constant (the Boltzmann constant).
-
Liquid Phase (Intra- and Intermolecular Forces)
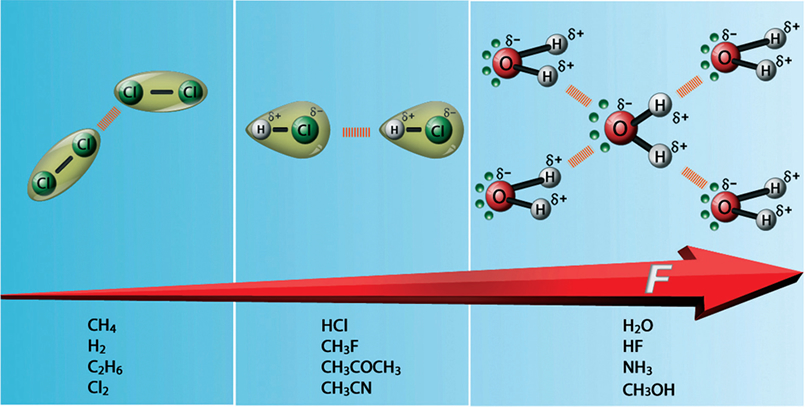
Van Der Waal's forces (weak) and hydrogen bonding (strong). London forces between Cl2 molecules, dipole-dipole forces between HCl molecules and H-bonding between H2O molecules. Note that a partial negative charge on an atom is indicated by ẟ- (delta negative), while a partial positive charge is indicated by ẟ+ (delta positive). Notice that one H2O molecule can potentially form 4 H-bonds with surrounding molecules which is highly efficient. The preceding is one key reason that the boiling point of water is higher than that of ammonia, hydrogen fluoride, or methanol.
- Surface Tension
- PE is directly proportional to the surface area (A)
- PE = gA; g = surface tension
- g = F/l; F = force of contraction of surface; l = length along surface
(a) cohesive > adhesive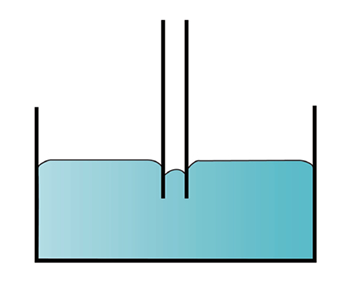 (b) adhesive > cohesive
(b) adhesive > cohesive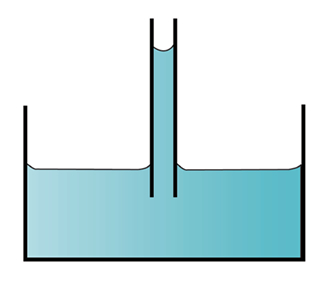
Solution Chemistry
- Vapor-Pressure Lowering (Raoult's Law)
where P = vapor pressure of solution
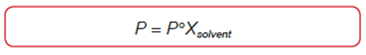
P° = vapor pressure of pure solvent (at the same temperature as P)
- Osmotic Pressure
where R = gas constant per mole
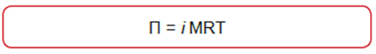
T = temperature in degrees K and
M = concentration of solute (mole/liter)
i = Van't Hoff factor
- Boiling-Point Elevation and Freezing-Point Depression
ΔTB=iKbm
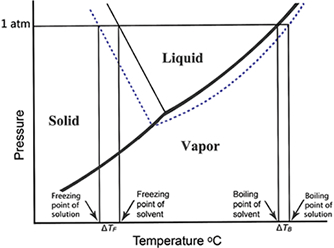
Phase diagram of water demonstrating the effect of the addition of a solute
ΔTF=iKFm
- Ions in Solution
- Ions that are positively charged = cations; ions that are negatively charged = anions
- Mnemonic: anions are negative ions
- The word "aqueous" simply means containing or dissolved in water
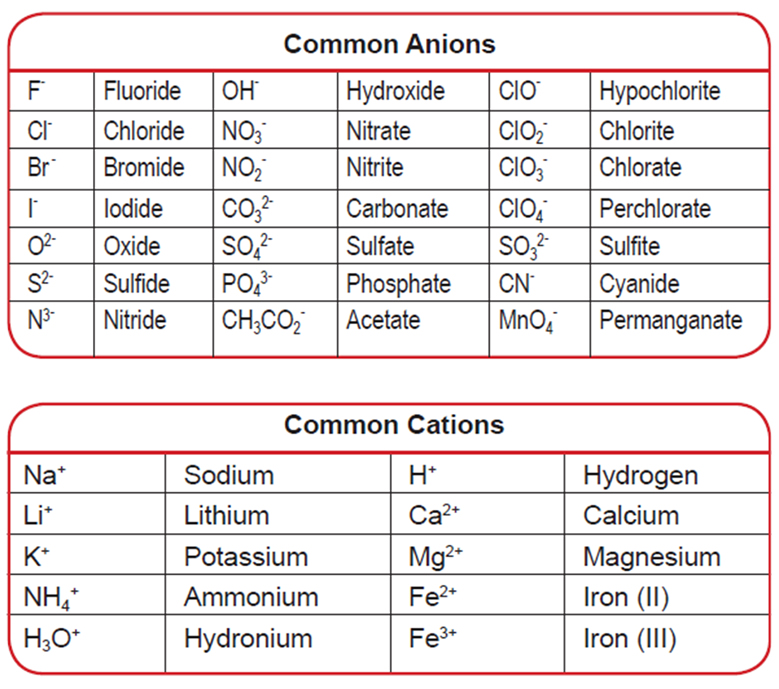
Common Anions and Cations
- Units of Concentration
- Molarity (M): moles of solute/liter of solution (solution = solute + solvent)
- Normality (N): one equivalent per liter
- Molality (m): one mole/1000g of solvent
- Molal concentrations are not temperature-dependent as molar and normal concentrations are
- Density (ρ): Mass per unit volume at the specified temperature
- Osmole (Osm): The number of moles of particles (molecules or ions) that contribute to the osmotic pressure of a solution
- Osmolarity: osmoles/liter of solution
- Osmolality: osmoles/kilogram of solution
- Mole Fraction: amount of solute (in moles) divided by the total amount of solvent and solute (in moles)
- Dilution: MiVi = MfVf
- Solubility Product Constant, the Equilibrium Expression
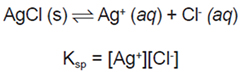
Because the Ksp product always holds, precipitation will not take place unless the product of [Ag+] and [Cl-] exceeds the Ksp.
- Solubility Rules
- All salts of alkali metals are soluble.
- All salts of the ammonium ion are soluble.
- All chlorides, bromides and iodides are water soluble, with the exception of Ag+, Pb2+, and Hg22+.
- All salts of the sulfate ion (SO42-) are water soluble with the exception of Ca2+, Sr2+, Ba2+, and Pb2+.
- All metal oxides are insoluble with the exception of the alkali metals and CaO, SrO and BaO.
- All hydroxides are insoluble with the exception of the alkali metals and Ca2+, Sr2+, Ba2+.
- All carbonates (CO32-), phosphates (PO43-), sulfides(S2-) and sulfites (SO32-) are insoluble, with the exception of the alkali metals and ammonium.
Acids & Bases
Acids
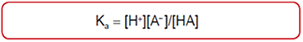
STRONG WEAK Perchloric HClO4
Chloric HClO3
Nitric HNO3
Hydrochloric HCl
Sulfuric H2SO4
Hydrobromic HBr
Hydriodic HI
Hydronium Ion H3O+
Hydrocyanic HCN
Hypochlorous HClO
Nitrous HNO2
Hydrofluoric HF
Sulfurous H2SO3
Hydrogen Sulfide H2S
Phosphoric H3PO4
Benzoic, Acetic and other Carboxylic Acids
Water Dissociation
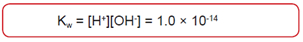
Salts of Weak Acids and Bases
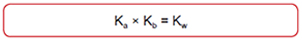
Buffers

Bases

- Strong bases include any hydroxide of the group 1A metals
- The most common weak bases are ammonia and any organic amine.
- Conjugate Acid-Base Pairs
- The acid, HA, and the base produced when it ionizes, A-, are called a conjugate acid-base pair.
- The pH Scale
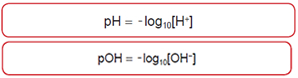
at 25°C, pH + pOH = 14.0
Thermodynamics
The First Law of Thermodynamics
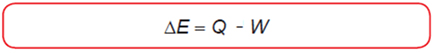
- heat absorbed by the system: Q > 0
- heat released by the system: Q < 0
- work done by the system on its surroundings: W > 0
- work done by the surroundings on the system: W < 0
Temperature Scales
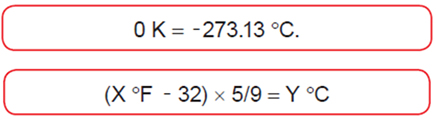
State Functions
W can be determined experimentally by calculating the area under a pressure-volume curve
Work Heat Changes in internal energy 1st tranf. w 0 -w 2nd transf. W = w +q q -w
Enthalpy & Thermochemistry
- Heat of Reaction: Basic Principles
- A reaction during which heat is released is said to be exothermic (ΔH is negative).
If a reaction requires the supply of a certain amount of heat it is endothermic (ΔH is positive).
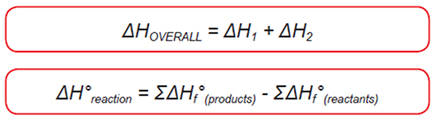
Bond Dissociation Energies and Heats of Formation
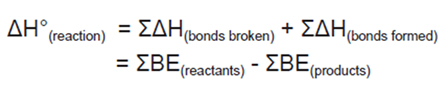
Calorimetry
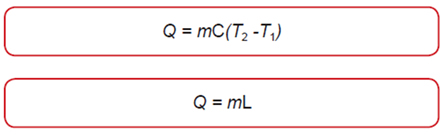
The Second Law of Thermodynamics
- For any spontaneous process, the entropy of the universe increases which results in a greater dispersal or randomization of the energy (ΔS > 0).
Entropy

Free Energy
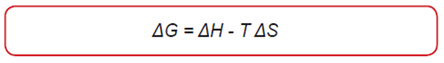
- A reaction carried out at constant pressure is spontaneous if: ΔG < 0
- It is not spontaneous if: ΔG > 0
- It is in a state of equilibrium (reaction spontaneous in both directions) if: ΔG = 0
Rate Processes in Chemical Reactions
Dependence of Reaction Rates on Concentration of Reactants
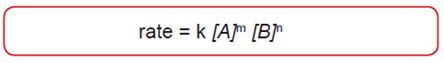
- [ ] is the concentration of the corresponding reactant in moles per liter
- k is referred to as the rate constant
- m is the order of the reaction with respect to A
- n is the order of the reaction with respect to B
- m+n is the overall reaction order
Dependence of Reaction Rates upon Temperature
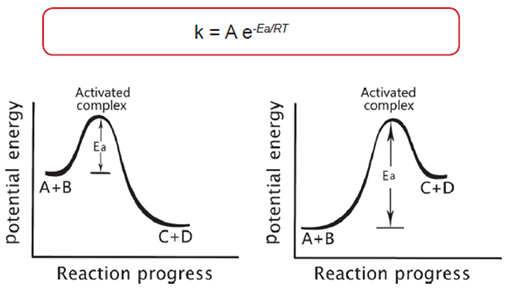
Potential Energy Diagrams: Exothermic vs. Endothermic Reactions
Catalysis
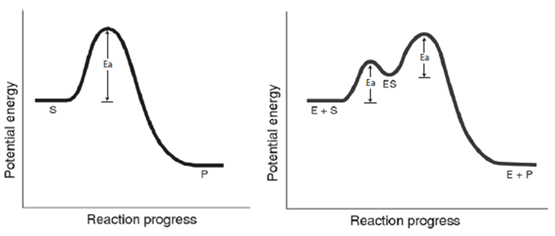
Potential Energy Diagrams: Without and With a Catalyst
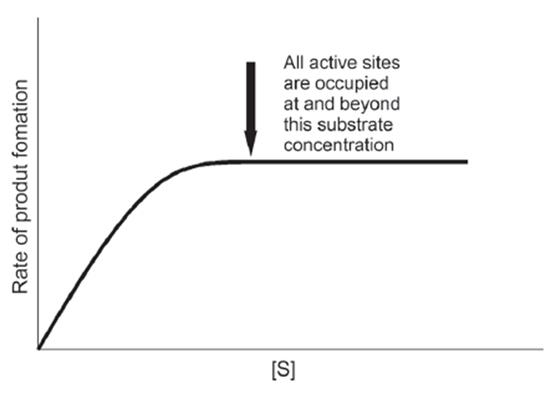
- Equilibrium in Reversible Chemical Reactions
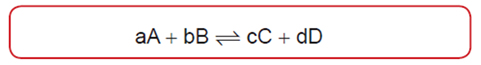
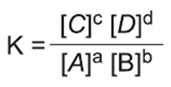
{Note: Catalysts speed up the rate of reaction without affecting Keq}
- Le Chatelier's Principle
- Le Chatelier's principle states that whenever a perturbation is applied to a system at equilibrium, the system evolves in such a way as to compensate for the applied perturbation.
Relationship between the Equilibrium Constant and the Change in the Gibbs Free Energy

Electrochemistry
Dependence of Reaction Rates on Concentration of Reactants
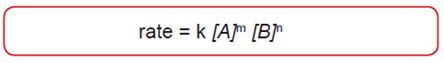
- [ ] is the concentration of the corresponding reactant in moles per liter
- k is referred to as the rate constant
- m is the order of the reaction with respect to A
- n is the order of the reaction with respect to B
- m+n is the overall reaction order
Dependence of Reaction Rates upon Temperature

Potential Energy Diagrams: Exothermic vs. Endothermic Reactions
Catalysis

Potential Energy Diagrams: Without and With a Catalyst

- Equilibrium in Reversible Chemical Reactions
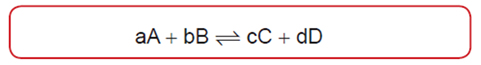
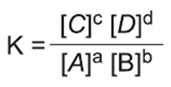
{Note: Catalysts speed up the rate of reaction without affecting Keq}
- Le Chatelier's Principle
- Le Chatelier's principle states that whenever a perturbation is applied to a system at equilibrium, the system evolves in such a way as to compensate for the applied perturbation.
Relationship between the Equilibrium Constant and the Change in the Gibbs Free Energy

Editorial Note: This content and the accompanying practice materials were developed and vetted by the Gold Standard MCAT Editorial Team. Our team—consisting of experienced medical educators, physicians, and subject matter specialists—is dedicated to providing the most accurate, representative prep materials and online courses for U.S. and Canadian premedical students.